⎯⎯⎯ Captivate RTSM
Randomization and Trial Supply Management system



Modern Interventional Studies
Robust Randomization Strategies
Captivate RTSM is a web-based Randomization and Trial Supply Management module designed to support a wide range of randomization strategies while maintaining operational simplicity, compliance, and audit readiness.
Efficient Trial Supply Management
Built for sponsors and CROs running interventional studies, Captivate RTSM delivers robust randomization capabilities without the overhead and rigidity of traditional IRT systems.
Unified Clinical Data Environment
Designed to integrate seamlessly with the Captivate clinical data platform, Captivate RTSM enables study teams to configure and manage randomization schemes, maintain blinding, and track investigational kits within a single, unified environment.
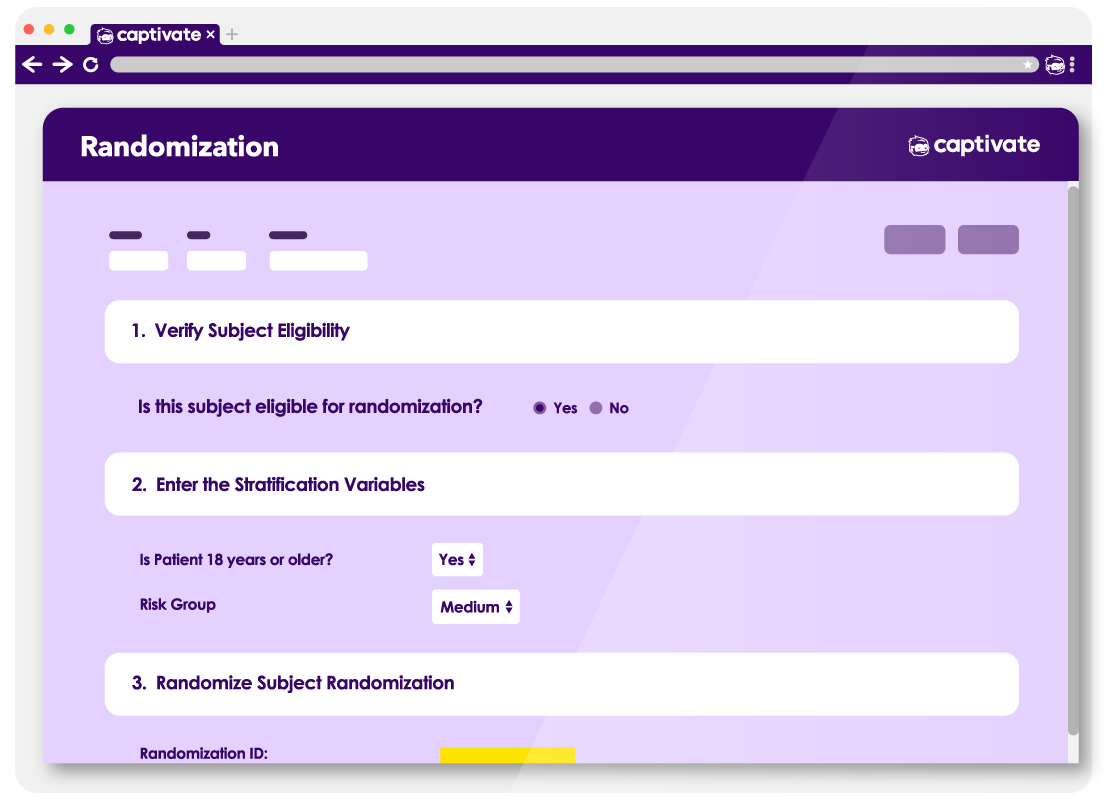
Simple Randomization
Easily configure simple randomization logic for straightforward study designs.
Stratified Randomization
Support for randomization across protocol-defined factors to ensure balance.
Permuted Block
Configurable block sizes for permuted block randomization strategies.
Dynamic Randomization
Support for dynamic randomization to maintain balance across treatment arms in real-time.
Real-Time Enforcement
Enforcement of randomization rules at the point of assignment for compliance.
Centralized Management
Logic managed centrally for consistent application across all sites and subjects.
Single & Double Blind
Comprehensive support for both single- and double-blinded designs
Role-Based Access
Protects blind integrity through controlled access levels
Emergency Workflows
Controlled and logged emergency unblinding workflows
Complete Audit Trails
Full traceability for all unblinding events during the study

Identification and Tracking
Centralized Setup
Define kit identifiers centrally for efficient oversight across the study.
Automated Assignment
Assignment of kits based directly on randomization results.
Lifecycle Tracking
Complete tracking of kit status throughout the entire study lifecycle.
Supply Visibility
Visibility into used, assigned, and available kits for sites and study teams.
Secure Browser Access
Secure, web-based interaction with the RTSM module
Guided Assignments
Step-by-step workflows for randomization and kit assignment
Immediate Confirmation
Real-time confirmation for all randomization actions
Subject History
Clear visibility into individual subject and kit history

The Captivate Platform
Shared Context
Shared subject and study context with Captivate EDC.
Unified Audit Trails
Consistent tracking across randomization, kits, and clinical data.
Simplified Reporting
Exports and reports that include critical randomization metadata.
Reduced Effort
Minimized reconciliation effort across clinical systems.

Compliance with 21 CFR Part 11 and Annex 11
Secure system access and data handling
Comprehensive audit trails for randomization and blinding
Documented traceability for inspections and monitoring
Identification and Tracking
More Flexible
Avoids the rigidity of systems that require extensive customization.
More Structured
Highly auditable alternative to manual or spreadsheet-based randomization.
No Extra Overhead
Designed to support study needs without unnecessary operational burden.
Choose Captivate
Robust Support
For multiple randomization methods
Blinding Controls
Built-in support for unblinding
Integrated Kit Tracking
Aligned with randomization outcomes
Unified Workflows
Consistent context with Captivate EDC
Inspection-Ready
Auditability without unnecessary complexity
Ready to Learn More?
Discover how Captivate can support your clinical research needs. Our team is here to answer questions and show you what’s possible.

