Captivate eTMF
Effortless Study Document Management
Trial Master File solution for effortless document management
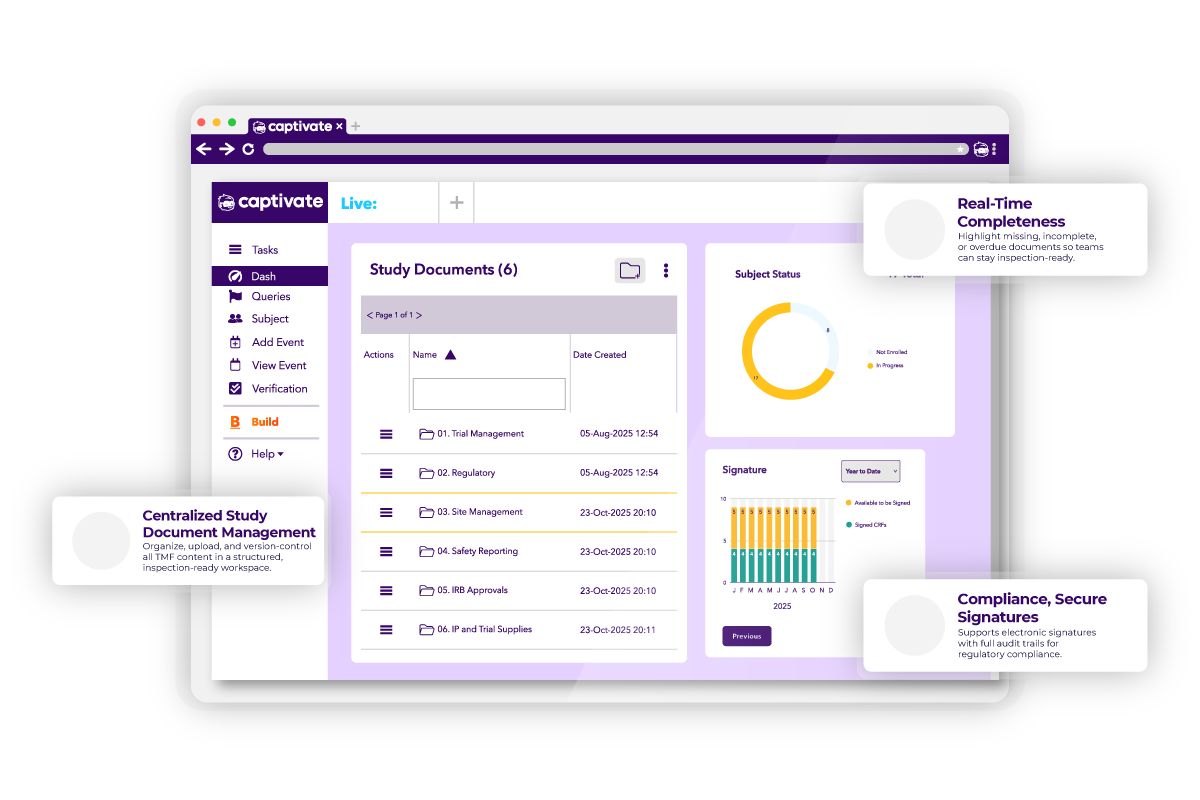
Captivate eTMF (Electronic Trial Master File) is designed to help studies and sponsors efficiently organize and manage clinical trial documentation, ensuring smooth submissions at the conclusion of the data collection cycle. Built on the robust Captivate platform, it simplifies the complexities of trial documentation while maintaining the highest standards of privacy, security, and scalability.
Access & Security
- Permission-based access control restricts document viewing and management to authorized personnel only
- Role-based design with focus on user roles and permissions
- Clear audit trail functionality for all document activities
Document Management
- User-friendly workflows for folder creation, editing, and organization
- Version control tracking document iterations
- Upload/download capabilities with expiration date management
- Document movement and organization tools
Compliance & Reporting
- Global regulatory compliance: 21 CFR Part 11, HIPAA, GDPR, and Annex 11
- Comprehensive reporting generating overviews of all study documentation
- Scalable storage solutions with pre-signed links for secure file transfers

