⎯⎯⎯ Captivate Coder



for Clinical Trials
Standardized Medical Coding
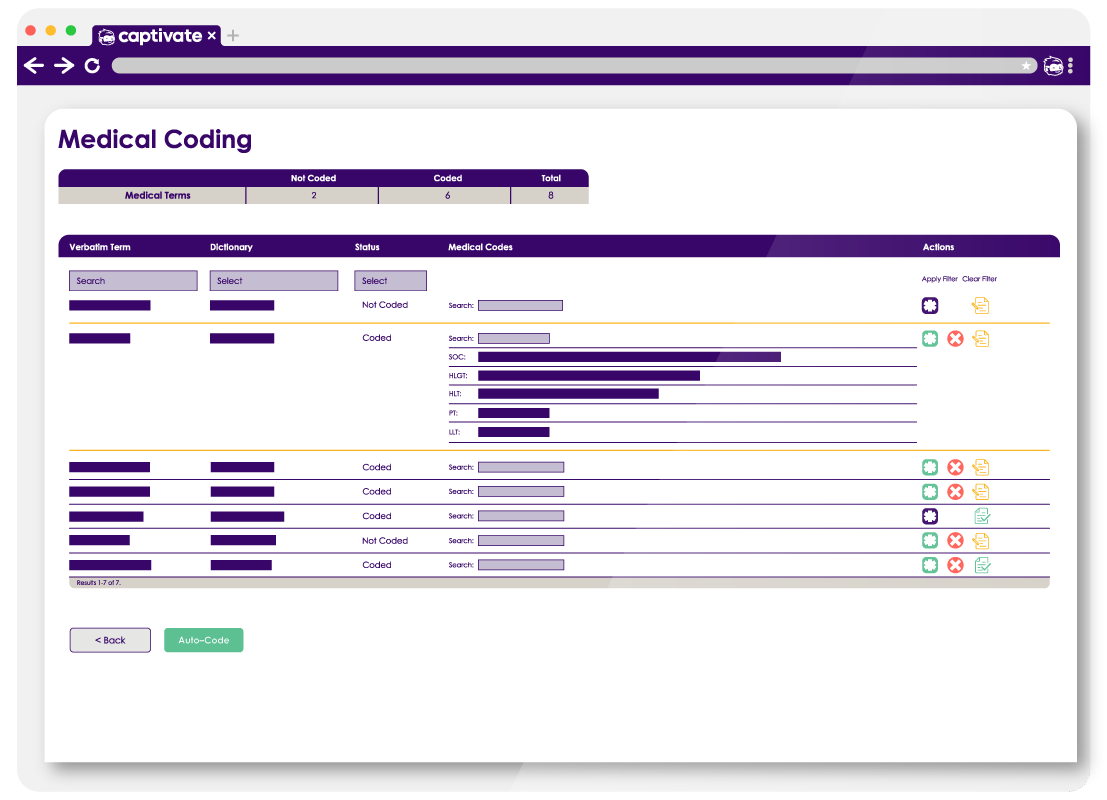
Captivate Coder is a flexible, integrated medical coding solution designed to help sponsors, CROs, and clinical data managers standardize adverse events, concomitant medications, and medical history terms using recognized medical dictionaries.
Seamless Platform Integration
Built to work within the Captivate clinical data platform, Captivate Coder provides structured, traceable, and compliant coding workflows that support data quality, analysis readiness, and regulatory expectations.
Automated Efficiency & Quality
By combining intuitive workflows with configurable automation and quality controls, Captivate Coder helps teams reduce manual effort, improve consistency, and maintain full auditability throughout the coding lifecycle.
Coding
Global Dictionary Support
Widely adopted dictionaries including MedDRA, WHO-Drug, ICD, and CTCAE.
Configurable Versions
Dictionary versions based on study requirements to support diverse protocols.
Comprehensive Mapping
Term mapping for adverse events, medications, medical history, and coded fields.
Hierarchical Structures
Support for hierarchical term structures to preserve critical context and consistency.
Balance automated suggestions with structured human review to support both efficiency and accuracy. Reduce manual effort while improving consistency.
Automated Suggestions
Initial proposals based on dictionary matches and term patterns
Pattern Matching
Dictionary-aware pattern matching to improve first-pass accuracy
Custom Coding Rules
Rule-based suggestions to surface preferred term mapping
Real-Time Feedback
Continuous feedback loops for coding specialists during review

Role-Based Workflows
Secure review and approval workflows tied to specific study permissions.
Version History
Complete tracking for coded terms and change history for every decision.
Adjudication Support
Commenting and rationale capture to support thorough coding adjudication.
Searchable Audit Trails
Fully traceable logs tied to all coding activities and decisions.
Manage clinical data and coded terms within a unified ecosystem. Eliminate translation gaps and support analysis quality.
Direct CRF Access
Code CRF terms directly without the need for data export
Shared Context
Shared subject and event context between clinical and coded data
Unified Reporting
Dashboards that include both source and coded clinical data
Analysis Readiness
Simplified exports for regulatory submission readiness

More Integrated
No more disconnected coding spreadsheets or manual workflows.
More Structured
Better traceability than point solutions that lack audit trails.
Supports Any Study
Designed to support studies of varying size and complexity.

Designed to meet regulatory expectations and support disciplined quality processes without unnecessary complexity.
Compliance with 21 CFR Part 11, Annex 11, and GDPR
Secure, role-based system access tied to permissions
Detailed audit trails for coding decisions and workflows
Configurable dictionary versioning for transparency
Choose Captivate
Standards-Aligned
Application of global medical dictionaries
Balanced Automation
Assisted coding with controlled human review
Integrated Workflows
Unified directly with Captivate EDC
Traceable Histories
Full audit trails for quality oversight
Configurable Rules
Dictionary versions to support diverse protocols
Ready to Learn More?
Discover how Captivate can support your clinical research needs. Our team is here to answer questions and show you what’s possible.

